A Natural Compound that May Help the Brain Heal
N,N-Dimethyltryptamine, (DMT), is a naturally occurring tryptamine producing effects similar to LSD, ketamine, psilocybin and psilocin. DMT occurs naturally in many plant species and animals including humans and has been used in religious ceremonies as a traditional spiritual medicine by indigenous peoples in the Amazon basin. DMT can also be synthesized in a laboratory.
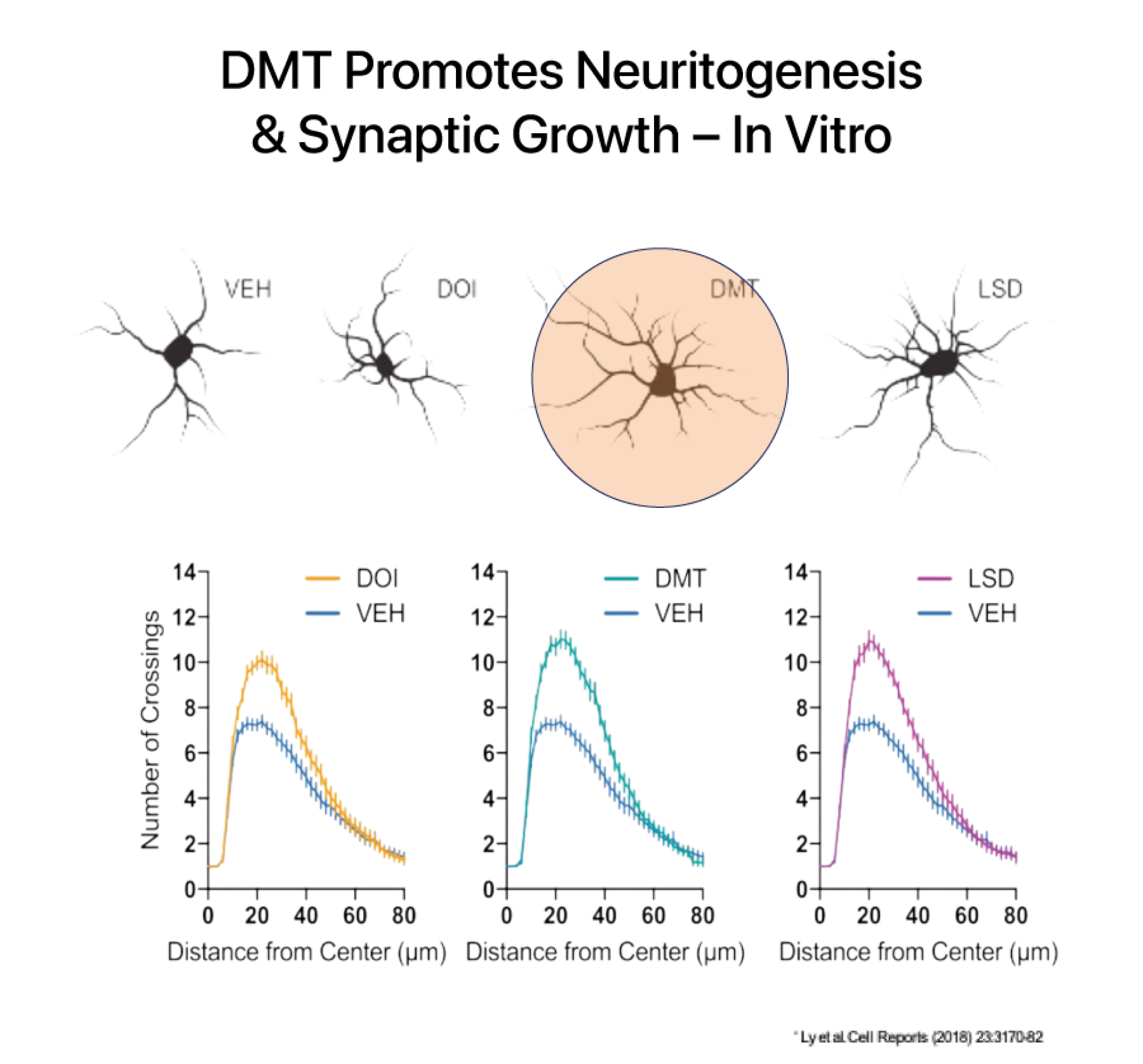
DMT has a rapid onset, intense effects, and a relatively short duration of action at high doses. At sub-hallucinogenic doses, DMT has been shown to induce and improve structural and functional neuroplasticity both in vitro and in preclinical murine models.
DMT increases brain derived neurotropic factor (BDNF) which is a key mechanism involved in healing the brain after an injury like a stroke. DMT is believed to activate pathways involved in forming neuronal connections and has been shown to increase the number of dendritic spines on cortical neurons. Dendritic spines form synapses (connections) with other neurons and are a critical site of molecular activity in the brain.
A Phase 1 DMT trial has recently been completed at the Centre for Human Drug Research in Leiden, The Netherlands. The study met all safety and tolerability criteria, including at the highest dose, which remained below the psychedelic threshold. We are now moving forward with investigating an intravenous sub-hallucinogenic dose of DMT in acute ischemic stroke patients, in a planned Phase 2a DMT stroke study to take place in Hungary in Q3 2025.
This planned randomized, double-blind, placebo-controlled Phase 2a DMT study of 40 ischemic stroke patients will focus on safety as its primary endpoint. Importantly, secondary endpoints will assess the potential impact of sub-psychedelic doses of DMT on cognitive functions, such as vision, hearing, sound, aphasia (a person’s ability to understand, speak, read, and write), motor function, and the size of the area of brain damage (infarct volume). A second Phase 2a study for traumatic brain injury (TBI) will be planned as well.